
Discover the optimal statin dosing time: Evening for short-acting (simvastatin, lovastatin) to match nocturnal cholesterol peaks; flexible anytime for long-acting (atorvastatin, rosuvastatin). Evidence-based guide maximizes LDL reduction and adherence.
Statins are the most widely prescribed anti-hypercholesterolemic drugs for lowering cholesterol levels and preventing atherosclerotic cardiovascular disease. Although their efficacy is well established, the optimal timing of administration remains an area of clinical interest. This article evaluates the mechanism of action of statins, their types, and available clinical evidence to determine the most effective timing for administration.
Statins and Their Mechanism
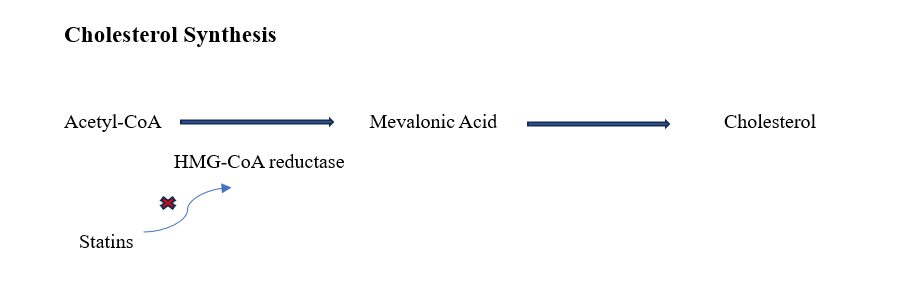
Statins are also known as HMG-CoA reductase inhibitors because they exert their effect by competitively inhibiting HMG-CoA reductase, the rate-limiting enzyme in the mevalonate pathway responsible for hepatic cholesterol biosynthesis. By blocking this enzyme, statins reduce intracellular cholesterol levels and upregulate LDL receptors on hepatocytes, thereby increasing the clearance of circulating LDL-cholesterol and lowering plasma LDL-cholesterol and total cholesterol levels.
Cholesterol synthesis is not constant throughout the day; it is regulated by circadian rhythm. Hepatic HMG-CoA reductase activity peaks during the late evening and early nighttime hours, particularly when caloric intake is low. This nocturnal increase in cholesterol production provides a mechanistic rationale for aligning statin dosing with the evening or nighttime, so that peak plasma drug concentrations coincide with peak enzyme activity and maximize LDL-cholesterol suppression.
Different Kinds of Statins
Statins are categorized based on their half-life into short-acting and long-acting agents. They differ in potency, half-life, and metabolism, which in turn influence how timing affects their lipid-lowering efficacy.
Short-acting statins, such as simvastatin, lovastatin, and fluvastatin, have relatively short half-lives (typically around 1–3 hours) and are most effective when their peak activity coincides with the body’s peak cholesterol synthesis, which occurs at night. Therefore, these agents are generally recommended for evening or bedtime dosing. Lovastatin, in particular, should be taken with the evening meal to improve its absorption.
In contrast, long-acting statins such as atorvastatin and rosuvastatin have half-lives of approximately 14–20 hours or longer, allowing sustained inhibition of HMG-CoA reductase over a 24-hour period regardless of dosing time. As a result, their effectiveness is less dependent on the timing of administration.
These pharmacokinetic differences explain why short-acting and long-acting statins respond differently to morning versus evening dosing.
So, what is the Right Time to Take Statins?
The timing of statin administration, whether in the morning or evening, may influence their pharmacokinetic and pharmacodynamic profiles, particularly for short-acting agents.
A 2022 systematic review and meta-analysis of randomized trials found that evening dosing of statins resulted in greater reductions in LDL-cholesterol and total cholesterol compared to morning dosing, with an average difference of approximately 6 mg/dL for LDL-cholesterol and a similar magnitude for total cholesterol. The effect was most pronounced for short half-life statins, where nocturnal administration reduced LDL-cholesterol by approximately 0.21 mmol/L and total cholesterol by 0.32 mmol/L more than morning dosing. However, these differences, although statistically significant, are modest and may not be clinically meaningful for all patients.
For short-acting statins, several clinical trials and meta-analyses consistently demonstrate that evening dosing produces greater lipid-lowering effects than morning dosing, reinforcing the recommendation that these agents should be taken at night.
In contrast, for long-acting statins such as atorvastatin and rosuvastatin, studies generally report no clinically meaningful difference in LDL-cholesterol reduction between morning and evening administration. This allows dosing time to be selected based primarily on patient preference, convenience, and adherence.
Graded Approach to Statin Dosing Timing
For short-acting statins, evening or bedtime dosing should be prioritized to maximize lipid-lowering efficacy.
For long-acting statins, patients may take the medication in the morning or evening based on their daily routine, comfort, and other concurrent medications.
Consistency in timing is often underappreciated. Taking the statin at the same time each day helps maintain stable drug levels and predictable lipid control. In clinical practice, adherence and regularity are more important than small differences in timing, particularly for long-acting agents.
Special consideration may be needed in certain situations, such as shift workers, patients with complex medication schedules, or those experiencing statin-associated side effects, where individualized timing may improve tolerability and adherence.
Summary
The optimal time to take statins depends primarily on the type of statin and the patient’s routine.
For short-acting statins (e.g., simvastatin, fluvastatin), evening or bedtime dosing is preferred because it aligns with the nocturnal peak in cholesterol synthesis and results in slightly greater reductions in LDL-cholesterol and total cholesterol.
For long-acting statins (e.g., atorvastatin, rosuvastatin), dosing time is more flexible, as morning and evening administration provide similar lipid-lowering effects. In these cases, the focus should shift toward ensuring consistent daily use and long-term adherence.
In clinical practice, the most effective strategy is to match the statin’s pharmacokinetic profile with the patient’s lifestyle: evening dosing for short-acting statins and a patient-tailored dosing time for long-acting statins, while consistently emphasizing adherence and persistence with therapy.

About the Writer
Dr. Preethi Putti, PharmD (LinkedIn Profile) is a pharmaceutical researcher with experience in market research, drug development pipelines, and regulatory analysis. She specializes in evaluating clinical trials and translating complex scientific data into clear, structured medical content. She is currently expanding her expertise in artificial intelligence in healthcare, with interests in digital health and clinical research innovation.
