30 June 2025
Category: Pharmaceutical Market Analysis I Drug Comparison & Reviews I
Obesity Therapeutics
Written by:
Priyanka Khamkar, MPharm, and
Vikas Londhe, MPharm
Reviewed By:
Pharmacally Editorial Team
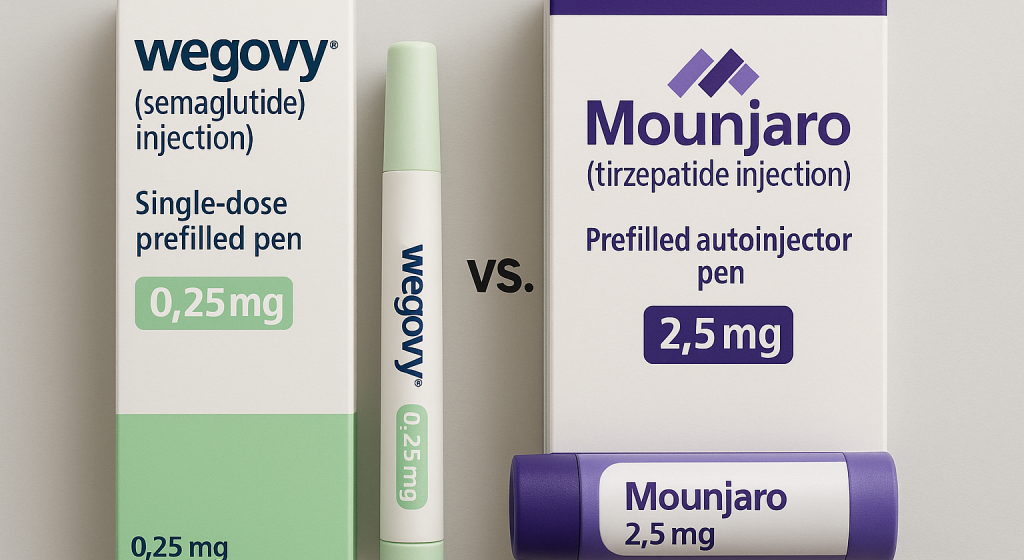
Wegovy (Semaglutide), the widely acclaimed GLP-1 agonist for weight loss, has officially launched in India by Danish pharmaceutical giant Novo Nordisk, following its overseas success. Wegovy’s launch has set the stage for a direct face-off with Eli Lilly’s Mounjaro (Tirzepatide), which entered the Indian market just three months earlier.
India is now home to the second-largest number of adults living with diabetes; obesity is also on the rise due to interdependent metabolic pathways, poor dietary habits, and increasingly sedentary urban lifestyles. Earlier in India, obesity was considered to be a lifestyle issue, and spending money on anti-obesity drugs was never on the priority list. But as the awareness about obesity and its associated health conditions is increased due to social media, physician counseling, the rise of nutritional health experts, and increased link of obesity with other ailments, it is now being recognized as a chronic, relapsing disease with serious health consequences, including cardiovascular disease, type-2 diabetes, and certain cancers.
Mounjaro, launched earlier this year in March, has already gained a foothold due to its superior weight loss efficacy (up to 23%) and promising results in both diabetes and obesity treatment. In contrast, Wegovy, which got approval in June 2025, has earned widespread global recognition for its effectiveness in achieving 15–20% weight loss and its cardiovascular benefits.
Wegovy is now available in major Indian cities, offering a new option for patients struggling with obesity and its associated health burdens. Its entry indicated not just another medicine option apart from Mounjaro, but a broader shift in India’s healthcare arena, where obesity is finally being addressed with serious medical intervention.
Drug Profiles & Mechanisms
Wegovy (semaglutide)
- Type: Once-weekly GLP-1 receptor agonist
- Mechanism of Action: Mimics glucagon-like peptide-1 (GLP-1), a natural satiety hormone. It reduces appetite, slows gastric emptying, and enhances insulin secretion, leading to reduced calorie intake and improved metabolic control.
- Approved Indications: Approved in India as the first obesity-specific medication with a clear indication for chronic weight management
- Also approved for cardiovascular risk reduction in overweight/obese individuals with established heart disease (based on the the SELECT trial).
- Clinical Trials & Efficacy: STEP trials (Semaglutide Treatment Effect in People with Obesity): Demonstrated average weight loss of 15%, with some patients reaching up to 20% at higher doses.
- Indian data indicates that 1 in 3 patients may achieve 20% weight loss with consistent high-dose use.
Mounjaro (Tirzepatide)
- Type: Once-weekly dual GIP and GLP-1 receptor agonist
- Mechanism of Action: Acts on both glucose-dependent insulinotropic polypeptide (GIP) and GLP-1 receptors, offering synergistic metabolic effects. It improves insulin sensitivity, reduces appetite, enhances satiety, and facilitates significant fat loss.
- Approved Indications: Initially approved in India for type 2 diabetes, now also approved for chronic weight management. In June 2025, India approved the Mounjaro KwikPen covering full doses (2.5–15 mg) for easier self-administration.
- Clinical Trials & Efficacy: SURMOUNT trials, particularly SURMOUNT-1 and SURMOUNT-5, showed average weight loss of 20–22.5%, outperforming semaglutide in head-to-head comparisons.
- Recognized for offering one of the highest weight reductions ever seen in obesity pharmacotherapy.
Launch Timeline
- Mounjaro launched in India in March 2025 via vials.
- Mounjaro KwikPen launched in June 2025.
- Wegovy launched on June 24, 2025
Dosage and Delivery Availability
Features | Wegovy Prefilled Pen | Mounjaro Vial | Mounjaro KwikPen (Approved in June 2025) |
Device Type | FlexPen (prefilled, single-use pen) | Vial for manual injection | KwikPen (prefilled, auto-injector) |
Dose Range | 0.25 mg – 2.4 mg | 2.5 mg – 5 mg | 2.5 mg – 15 mg |
Dosing Frequency | Once Weekly | Once Weekly | Once Weekly |
Wegovy is available in 5 different doses ranging from 0.25 mg, 0.5 mg, 1.0 mg, 1.7 mg, and 2.4 mg. It is available as a single-use FlexPen covering all step-up doses (0.25 mg to 2.4 mg), administered once a week.
The Mounjaro vial provides limited dose options available in two doses, 2.5 mg and 5 mg, which require manual preparation before injection.
Mounjaro, recently approved as a prefilled auto-injector in June 2025, named Mounjaro KwikPen, expands the dosing range up to 15 mg, enabling more convenient and auto-injector-based delivery, administered once weekly.
Pricing of Wegovy
Dose | Per Dose Pricing | Monthly Pricing |
0.25mg | Rs. 4366 | Rs. 17345 |
0.5mg | Rs. 4366 | Rs. 17345 |
1.0mg | Rs. 4366 | Rs. 17345 |
1.7mg | Rs. 6070 | Rs. 24280 |
2.4mg | Rs. 6503 | Rs. 26015 |
Pricing of Mounjaro
Dose | Per-Dose Pricing | Monthly Pricing |
2.5 mg | Rs. 3500 | Rs. 14000 |
5.0 mg | Rs. 4375 | Rs. 17500 |
Wegovy (semaglutide) is priced uniformly at ₹4,366 per dose for the initial lower doses (0.25 mg to 1.0 mg), with prices increasing for higher doses: 1.7 mg: ₹6,070 per dose (₹24,280/month); 2.4 mg (maintenance dose): ₹6,503 per dose (₹26,015/month)
Mounjaro (Tirzepatide) is more cost-effective in comparison: 2.5 mg (starter dose): ₹3,500 per dose (₹14,000/month); 5.0 mg (maintenance dose): ₹4,375 per dose (₹17,500/month)
Mounjaro offers a significantly more affordable monthly treatment, up to ₹8,500 less per month at maintenance doses, making it a cost-attractive option in India’s growing anti-obesity drug market. However, Wegovy is looking more expensive as compared to Mounjaro, but it is backed by robust global clinical trial data and proven long-term efficacy.
Market Growth and Patient Response
As Mounjaro is having a first-mover advantage, as it is launched in March 2025, it shows explosive growth in its first three months of sales. According to Business Today, data from March to May reveals a steady month-on-month increase in both value and volume sales. Revenue grew from ₹3.46 crore in March to ₹7.87 crore in April and ₹12.60 crore in May, with a cumulative total of ₹23.94 crore over three months. Similarly, unit sales increased from 11,640 units in March to 27,650 in April and further to 42,280 in May, bringing the total to over 81,000 units sold during this period. These figures reflect a strong ascending trend in physician prescribing and patient onboarding since the drug’s market entry in March.
Wegovy, which has now entered the Indian market, is expected to benefit from the same rising demand seen with Mounjaro. Previously, Wegovy faced global supply issues with limited production and distribution due to high demand worldwide. However, those supply problems have now mostly been resolved, specifically for the Indian market, allowing a more stable and consistent availability of the drug in India. This positions Wegovy to compete effectively and meet growing patient needs without the delays or shortages experienced earlier in other countries.
Market Size & Growth
As per the report of HORIZON Grand View Research, India’s anti-obesity medication market is ready for explosive growth. In 2023, the market generated revenue of approximately ₹1,525 crore (USD 183.4 million), and it is projected to reach around ₹21,790 crore (USD 2,619 million) by 2030. This growth represents an impressive compound annual growth rate (CAGR) of 46.2% from 2024 to 2030. The market surge is typically due to rising obesity rates, increasing awareness of related health risks, and a growing demand for effective medical treatments like Mounjaro and Wegovy. As medicine availability and accessibility improve and physician adoption increases, India is emerging as a major market for next-generation weight-loss therapies.
Strategic Battle: Wegovy vs Mounjaro in India
First-Mover Advantage
Eli Lilly gained an early lead by launching Mounjaro in March 2024, capturing physician attention and onboarding patients first. In response, Novo Nordisk quickly launches Wegovy, aiming to exploit the same momentum and not lose market share.
Device Convenience: Pens over Vials
Both companies now use pen devices instead of vials, which are more user-friendly, improve patient adherence, and support easy self-injection.
Efficacy Advantage
In clinical trials, Mounjaro (tirzepatide) showed up to ~23% weight loss, and Wegovy (semaglutide) showed 15–20% weight loss. This superior efficacy profile could tilt the market in favour of Mounjaro, especially for patients seeking maximum weight loss results.
Patent Expiry and Generic Entry
Wegovy (semaglutide) will lose patent protection in India by March 2026, opening the door for low-cost generics, potentially 60–90% cheaper
In contrast, Mounjaro (tirzepatide) remains patent-protected until around 2036, giving Lilly a longer pricing runway and protection from generic competition.
Final Take
India’s anti-obesity drug market is undergoing a crucial shift. Historically, obesity was not recognized as a medical condition in the country, and as a result, anti-obesity medications were often seen as non-essential or lifestyle products. Many patients were unwilling to invest in prescription-based weight loss treatments. However, for the first time, India is witnessing a competitive fight between two global pharmaceutical giants, Eli Lilly and Novo Nordisk, as they strive for dominance in this emerging therapeutic space. This competition signals a shift in perception, where obesity is increasingly considered a serious and treatable health condition, opening the door to more scientifically and medically proven weight management.
Elli Lilly’s Mounjaro currently holds a strategic edge with its superior efficacy (~23% weight loss) and longer patent protection until ~2036, making it well-suited for medically supervised, premium patients who prioritize results and can afford higher prices. Its early market entry and KwikPen device covering the full dose range further strengthen its position.
On the other hand, Novo Nordisk’s Wegovy brings a powerful mix of strong global brand equity and proven cardiovascular benefits, making it highly appealing, especially as obesity increasingly intersects with metabolic and heart health. Novo Nordisk’s accelerated India launch shows a clear intent to capture momentum, particularly in urban centers, before generics flood the market post-2026, when semaglutide’s patent expires.
References
Novo Nordisk launches weight-loss drug Wegovy in India to compete with Lilly’s Mounjaro, Reuters, June 24 2025, https://www.reuters.com/business/healthcare-pharmaceuticals/novo-nordisk-launches-blockuster-weight-loss-drug-wegovy-india-2025-06-24/
Novo Nordisk’s Wegovy debuts in India: How it works, who it’s for, and how much it costs, Times of India, June 27 2025, https://timesofindia.indiatimes.com/life-style/health-fitness/health-news/novo-nordisks-wegovy-debuts-in-india-how-it-works-who-its-for-and-how-much-it-costs/articleshow/122054770.cms
Novo Nordisk launches blockbuster weight-loss drug Wegovy in India; Check price, dosage and other details, The Economics Times, June 24 2025, https://economictimes.indiatimes.com/news/new-updates/novo-nordisk-launches-blockbuster-weight-loss-drug-wegovy-in-india-check-price-dosage-and-other-details/articleshow/122042130.cms?from=mdr
Highlights of prescribing information, Wegovy, https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/215256s007lbl.pdf
Highlights of prescribing information, Mounjaro, https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215866s000lbl.pdf
Lilly to launch Mounjaro pen in India to compete with Novo’s weight-loss drug Wegovy, Reuters, June 26 2026, https://www.reuters.com/business/healthcare-pharmaceuticals/lilly-launch-mounjaro-pen-india-compete-with-novos-wegovy-2025-06-26/
Eli Lilly’s Mounjaro hits Rs 24 crore sales in 3 months as adoption grows for diabetes and obesity in India, Business Today, 08 June 2025, https://www.businesstoday.in/industry/pharma/story/eli-lillys-mounjaro-hits-rs-24-crore-sales-in-three-months-as-adoption-grows-for-diabetes-and-obesity-in-india-479483-2025-06-08
Eli Lilly launches weight-loss drug Mounjaro in India, beats Novo Nordisk to major market, March 21 2025, https://www.reuters.com/business/healthcare-pharmaceuticals/eli-lilly-launches-weight-loss-drug-mounjaro-india-after-drug-regulator-approval-2025-03-20/
India Anti-Obesity Medication Market Size & Outlook, HORIZON Grand View Research, https://www.grandviewresearch.com/horizon/outlook/anti-obesity-medication-market/india