Argenx SE announces positive Phase 3 ADAPT OCULUS results for VYVGART (efgartigimod alfa) in ocular myasthenia gravis (oMG), meeting primary endpoint with significant MGII ocular score improvements vs placebo. The trial offers hope for oMG patients with diplopia/ptosis relief; full data pending medical meeting.
Written by: Pharmacally Medical News Desk
Argenx SE has announced positive topline results from the Phase 3 ADAPT OCULUS study of VYVGART (efgartigimod alfa and hyaluronidase-qvfc) in adults with ocular myasthenia gravis (oMG). The trial met its primary endpoint, demonstrating significant improvements in patient-reported ocular symptoms.
Luc Truyen, Chief Medical Officer of argenx, noted that ADAPT OCULUS marks the first registrational study designed specifically for a targeted therapy in ocular myasthenia gravis. He emphasized its role in addressing a historically under-studied need, advancing patient-centered development toward transformative options for all MG patients.
Study Results
The ADAPT OCULUS trial (NCT06558279) showed a statistically significant improvement in the Myasthenia Gravis Impairment Index (MGII) patient-reported outcome (PRO) ocular scores at Week 4, with a p-value of 0.012.
Patients treated with subcutaneous VYVGART achieved a mean 4.04-point improvement from baseline, compared to 1.99 points with placebo.
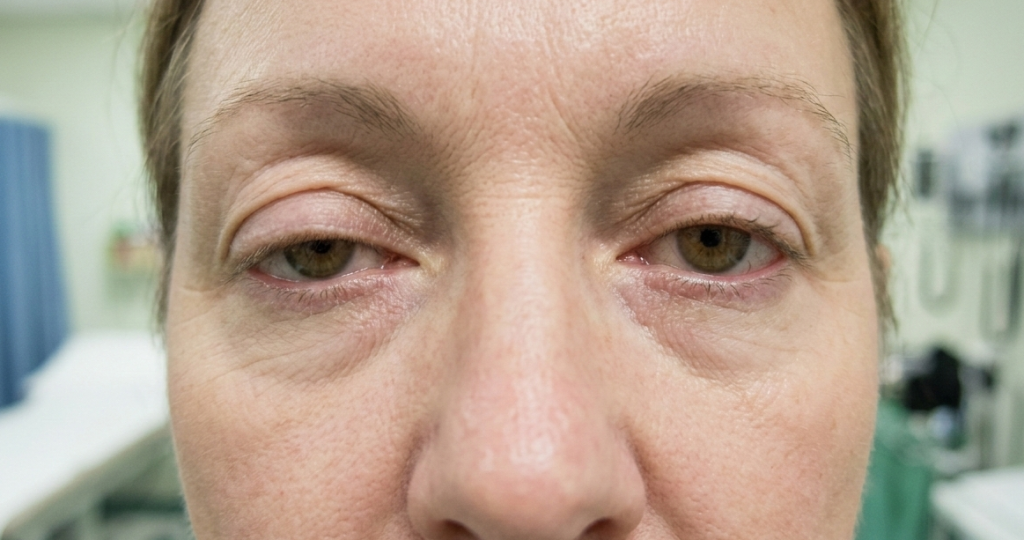
Key symptoms like diplopia (double vision) and ptosis (drooping eyelids) were markedly reduced.
Trial Design
This Phase 3, randomized, double-blind, placebo-controlled study enrolled 141 adults with MGFA Class I oMG across North America, Europe, and Asia-Pacific. Part A involved 1:1 randomization to four weekly subcutaneous injections of VYVGART or placebo, followed by a 4-week follow-up. Part B was an open-label extension. Participants, either AChR-Ab seropositive or seronegative, were on stable generalized MG treatments and had baseline MGII ocular scores ≥6.
Safety Profile
VYVGART was well-tolerated in oMG patients, aligning with prior studies and showing no new safety signals. For detailed adverse events, precautions, and warnings—including infections (e.g., respiratory tract infections in 33%), headache (32%), hypersensitivity reactions, and infusion-related reactions—refer to the full U.S. Prescribing Information. Common risks involve monitoring for infections and delaying treatment during active cases; live vaccines are not recommended during therapy.
For detailed information on adverse drug reactions (ADRs), precautions, and warnings for VYVGART including risks like infections (e.g., upper respiratory in 33%), headache (32%), infusion-related reactions, and hypersensitivity, consult the official U.S. Prescribing Information.
Key precautions involve monitoring for infections, avoiding live vaccines during treatment, and assessing for worsening MG symptoms.
Carolina Barnett-Tapia, M.D., Ph.D., Associate Professor of Medicine (Neurology) at the University of Toronto, highlighted how oMG severely disrupts vision, independence, and routine tasks like driving or working. She pointed out the absence of approved targeted treatments until now, with VYVGART’s trial improvements offering real hope to thousands affected.
Argenyx reported that full data will be presented at an upcoming medical meeting, supporting a planned sBLA submission to the FDA for oMG label expansion. VYVGART is currently approved for generalized MG in anti-AChR positive adults.
Reference
argenx Announces Positive Topline Results from Phase 3 ADAPT OCULUS Trial of VYVGART in Ocular Myasthenia Gravis, 26 February 2026, argenx | argenx Announces Positive Topline Results from Phase 3 ADAPT OCULUS Trial of VYVGART in Ocular Myasthenia Gravis
Prescribing Information, VYVGART, vyvgart-prescribing-information.pdf
A Study to Assess Efficacy and Safety of Efgartigimod PH20 SC in Adults With Ocular Myasthenia Gravis (ADAPT oculus), ClinicalTrials.gov ID NCT06558279, https://clinicaltrials.gov/study/NCT06558279
