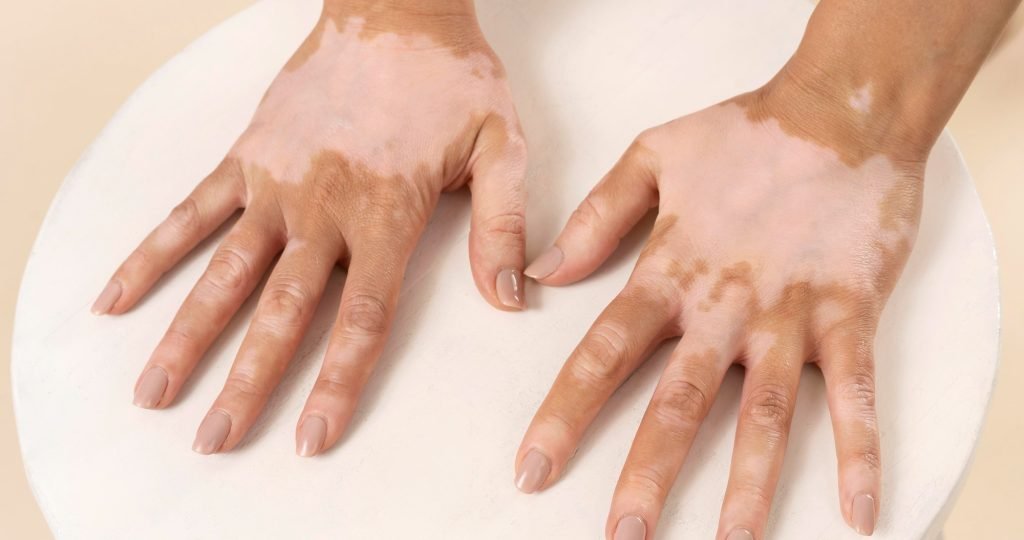
AbbVie submits FDA, EMA applications for RINVOQ (upadacitinib) in non-segmental vitiligo. Phase 3 Viti-Up data show significant repigmentation via JAK1 inhibition of IFN-γ pathways. Learn efficacy, safety, and vitiligo treatment advances.
Written By: Nikita Jha, BPharm
Reviewed By: Pharmacally Editorial Team
AbbVie has submitted regulatory applications to the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for upadacitinib (RINVOQ®; 15 mg once daily). The filings seek approval for treating adults and adolescents aged 12 years and older with non-segmental vitiligo (NSV), a chronic autoimmune skin disorder affecting pigmentation.
Supported by the Phase 3 Viti-Up clinical program, these submissions target patients eligible for systemic therapy. “Vitiligo’s unpredictability frustrates patients, and no systemic therapies exist today,” said Kori Wallace, MD, PhD, vice president and global head of immunology clinical development at AbbVie. “The Viti-Up studies address these gaps, reflecting our commitment to advancing vitiligo care.”
Phase 3 Viti-Up Topline Results
The Viti-Up program (NCT06118411) included two replicate double-blind, placebo-controlled trials enrolling 614 adults and adolescents with NSV across 90 global sites. Patients were randomized 2:1 to upadacitinib 15 mg once daily or placebo for 48 weeks, followed by a long-term open-label extension up to 160 weeks total.
Upadacitinib met both co-primary endpoints at week 48 with statistically significant repigmentation improvements:
Endpoint | Study 1 (Upadacitinib vs. Placebo) | Study 2 (Upadacitinib vs. Placebo) |
T-VASI 50 (≥50% total vitiligo area reduction) | 19.4% vs. 5.9% | 21.5% vs. 5.9% |
F-VASI 75 (≥75% facial vitiligo area repigmentation) | 25.2% vs. 5.9% | 23.4% vs. 6.9% |
Key secondary endpoints, including F-VASI 50, were also achieved.
Safety Profile
RINVOQ carries risks of serious infections, malignancies, cardiovascular events, and thrombosis. Adverse reactions vary by indication; consult full prescribing information for contraindications, drug interactions, and counselling: RINVOQ PI.
About RINVOQ and Vitiligo
RINVOQ (upadacitinib), an oral selective Janus kinase-1 (JAK-1) inhibitor, targets the core immune mechanisms driving non-segmental vitiligo (NSV) pathogenesis. In vitiligo, JAK pathways drive major mechanisms of melanocyte destruction by amplifying IFN-γ signalling and T-cell recruitment. By inhibiting JAK1, RINVOQ disrupts this cycle, reducing inflammation and promoting repigmentation.
Discovered and developed by AbbVie, RINVOQ (upadacitinib) is approved for multiple immune-mediated diseases, including inflammatory bowel disease such as ulcerative colitis and Crohn’s disease, and is under study in additional conditions including alopecia areata, hidradenitis suppurativa, systemic lupus erythematosus, and other inflammatory disorders.
NSV, comprising ~84% of vitiligo cases, causes symmetrical depigmented patches with psychosocial impacts. Current options focus on stabilization and repigmentation but lack approved systemic therapies. Approval for vitiligo is pending review.
References
AbbVie Submits Regulatory Applications to FDA and EMA for Upadacitinib (RINVOQ®) in Adults and Adolescents with Vitiligo, 03 February 2026, https://news.abbvie.com/2026-02-03-AbbVie-Submits-Regulatory-Applications-to-FDA-and-EMA-for-Upadacitinib-RINVOQ-R-in-Adults-and-Adolescents-With-Vitiligo
AbbVie Announces Positive Topline Results from Phase 3 Pivotal Studies Evaluating Upadacitinib (RINVOQ®) in Adults and Adolescents with Vitiligo, 29 October 2025, https://news.abbvie.com/2025-10-29-AbbVie-Announces-Positive-Topline-Results-from-Phase-3-Pivotal-Studies-Evaluating-Upadacitinib-RINVOQ-R-in-Adults-and-Adolescents-with-Vitiligo
A Study to Assess Adverse Events and Effectiveness of Upadacitinib Oral Tablets in Adult and Adolescent Participants with Vitiligo (Viti-Up), ClinicalTrials.gov ID NCT06118411, https://clinicaltrials.gov/study/NCT06118411
RINVOQ, Prescribing Information, https://www.rxabbvie.com/pdf/rinvoq_pi.pdf