MHRA issues Class 2 recall of Ramipril 10 mg capsules due to packaging error; some packs may contain 5 mg doses. Risk to patients assessed as low.
Written By: Mahathi Palivela, PharmD
Reviewed By: Pharmacally Editorial Team
A precautionary recall has been issued for a specific batch of Ramipril 10 mg capsules after a manufacturing error led to potential mispackaging of doses. The affected batch, GR174091 (expiry October 2026), was first distributed in May 2025.
According to the Medicines and Healthcare products Regulatory Agency (MHRA), some cartons labelled as 10 mg may contain blister strips of 5 mg capsules due to an error during secondary packaging at the manufacturing site.
The issue was identified following a patient complaint, prompting Crescent Pharma Limited to initiate a precautionary recall.
The Medicines and Healthcare products Regulatory Agency (MHRA) has classified the recall as a Class 2 recall, indicating a low risk to patients, noting that taking a lower dose of Ramipril for a short period is unlikely to cause immediate harm, as both 5 mg and 10 mg strengths are commonly used to treat high blood pressure, heart failure, and kidney disease.
Any impact from a reduced dose is expected to be gradual rather than acute.
Dr Alison Cave MHRA Chief Safety Officer advised patients prescribed 10 mg capsules to check the batch number on the outer carton and ensure the blister strips inside match the labelled strength. If a mismatch is found, patients should contact their dispensing pharmacy, while no action is required if the contents are correctly labelled.
Patients experiencing any adverse effects should seek medical advice and bring their medication packaging for review.
Healthcare professionals have been instructed to stop supplying the affected batch immediately, quarantine remaining stock, and return it to suppliers.
Pharmacies are also advised to identify and contact patients who may have received the impacted product, particularly those dispensed the medication between May 2025 and April 2026.
Any suspected adverse reactions should be reported through the MHRA’s Yellow Card Scheme.
References
Precautionary recall of blood pressure medication after manufacturing error – GOV.UK
Class 2 Medicines Recall: Crescent Pharma Limited, Ramipril 10mg capsules, EL(26)A/19 – GOV.UK

About the Writer
Mahathi Palivela is pursuing PharmD and has a strong interest in Clinical Pharmacy and Patient safety. She is passionate about handling and analyzing patient data, and translating clinical insights into clear, meaningful summaries. She aims to apply this interest in Medical Writing and Pharmacovigilance, focusing on improving patient outcomes through careful data interpretation and communication .
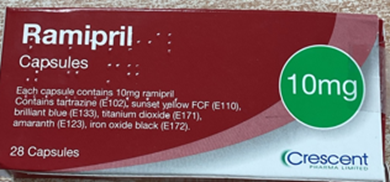


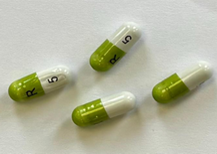
Actual Recalled Medicine Photographs
