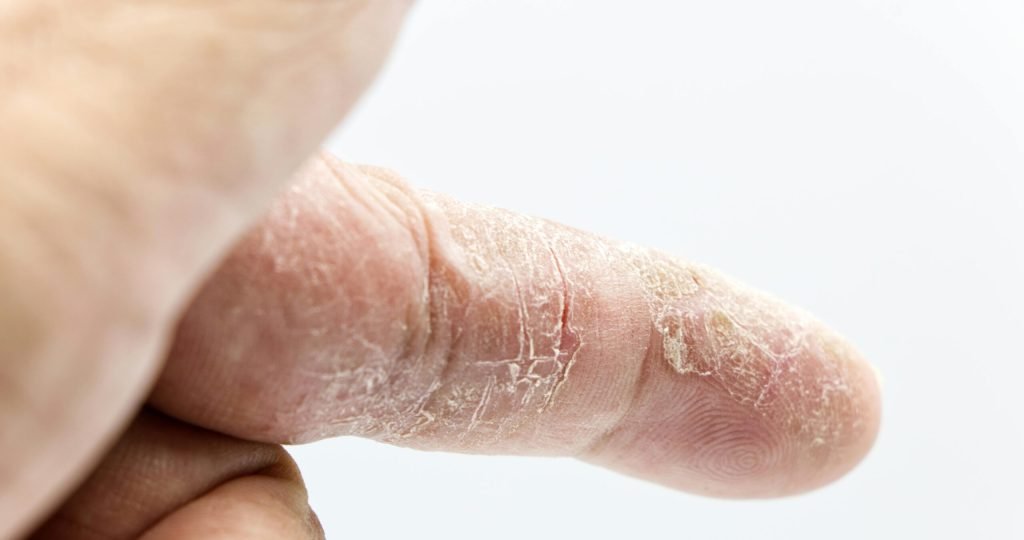
LEO Pharma’s ANZUPGO (delgocitinib) sNDA for adolescents with chronic hand eczema has been accepted by the FDA, supported by Phase 3 DELTA TEEN trial data.
Written By: Vennela Reddy, BPharm
Reviewed By: Pharmacally Editorial Team
LEO Pharma has advanced its effort to address a long-standing treatment gap in pediatric dermatology, as the U.S. Food and Drug Administration accepts a supplemental New Drug Application (sNDA) for ANZUPGO in adolescents aged 12–17 years with moderate to severe chronic hand eczema (CHE).
Expanding a Non-Steroidal Option Beyond Adults
The topical therapy, a non-steroidal pan-Janus kinase (JAK) inhibitor, is already approved in the United States for adult patients with CHE who have not responded adequately to topical corticosteroids or for whom such treatments are unsuitable. The new filing seeks to extend this indication to pediatric patients, a population currently lacking disease-specific approved therapy.
Company Highlights Unmet Pediatric Need
Bob Spurr, EVP and President, Region North America, LEO Pharma emphasized the unmet need in this group. The company noted that pediatric CHE has no approved targeted treatments and that expanding ANZUPGO’s label could help fill a critical gap.
Sophie Lamle, Executive Vice President, Development at LEO Pharma, highlighted that pediatric patients with chronic hand eczema currently lack specifically indicated treatment options in the U.S. She noted that the condition can affect patients during critical stages of personal development and added that the FDA’s acceptance of the filing marks an important step toward potentially providing a much-needed treatment option for this underserved population.
Phase 3 DELTA TEEN Trial Supports Submission
The submission is supported by results from the Phase 3 DELTA TEEN trial (NCT05355818), which evaluated twice-daily ANZUPGO application in adolescents with moderate to severe CHE. The study met its primary endpoint, demonstrating treatment success defined by clear or almost clear skin with at least a two-grade improvement from baseline at Week 16. Key secondary endpoints were also achieved. Detailed findings were presented at the European Academy of Dermatology and Venereology Congress 2025.
Disease Burden Remains Significant in Adolescents
Chronic hand eczema is a persistent and often debilitating condition characterized by inflammation, itching, pain, and visible skin damage such as fissures and scaling. In pediatric patients, the disease can interfere with daily activities and development, while treatment options remain limited due to concerns around long-term corticosteroid use.
Mechanism Offers Targeted Anti-Inflammatory Approach
ANZUPGO works by inhibiting JAK-STAT signaling pathways involved in inflammatory processes underlying CHE. Its non-steroidal mechanism offers a potential alternative to repeated corticosteroid use, which can carry safety and tolerability concerns with long-term application.
Global Regulatory Footprint Continues to Expand
Currently, ANZUPGO is approved for adult CHE across multiple regions, including the United States, European Union, and United Kingdom. The therapy is also under regulatory review in Europe for adolescent use, reflecting a broader global effort to expand access to targeted treatments for younger patients.
Next Step Toward First Pediatric CHE Therapy
With the FDA now formally reviewing the pediatric application, LEO Pharma moves closer to potentially introducing the first indicated therapy for adolescents with chronic hand eczema, marking a significant step toward addressing an underserved clinical need.
Reference

About the Writer
Vennela Reddy, B.Pharm is a pharmacy graduate with a keen interest in clinical research, pharmacovigilance, and medical writing, with a growing focus on publishable and scientific content development. In her words, she is passionate about translating complex medical data into clear, evidence-based communication.
