FDA issues a Complete Response Letter to Chiesi Global Rare Diseases for idebenone in Leber Hereditary Optic Neuropathy (LHON), requesting additional clinical data to support safety and efficacy.
Written By: Sana Khan BPharm
Reviewed By: Pharmacally Editorial Team
The U.S. Food and Drug Administration (FDA) has issued a Complete Response Letter (CRL) to Chiesi Global Rare Diseases for idebenone, an investigational therapy intended for adolescents and adults with Leber Hereditary Optic Neuropathy (LHON), the company announced.
In the letter, the agency requested additional clinical evidence from adequate and well-controlled studies to further demonstrate the safety and effectiveness of idebenone in patients with LHON.
The FDA also asked the company to provide clarification and updates related to chemistry, manufacturing, and nonclinical data. In addition, issues connected to a referenced Drug Master File must be resolved before the application can be reconsidered. The agency stated that no new clinical safety concerns were identified during the review.
The regulatory submission was supported by data from the pivotal RHODOS (NCT00747487) randomized double-blind, placebo-controlled trial and the LEROS (NCT02774005) Phase 4 open-label interventional study. Both studies evaluated the safety and efficacy of idebenone in adolescents and adults with LHON carrying primary mitochondrial DNA mutations.
“Although this outcome is not what we hoped for, we’re deeply grateful to the patients, families, and investigators whose dedication has enabled critical advancements in LHON research,” said Giacomo Chiesi, Executive Vice President of Chiesi Global Rare Diseases. He added that the company remains committed to supporting research efforts aimed at improving treatment options for people living with LHON.
Advocates from the LHON community also emphasized the continued need for research and collaboration to address the unmet medical need associated with the disease.
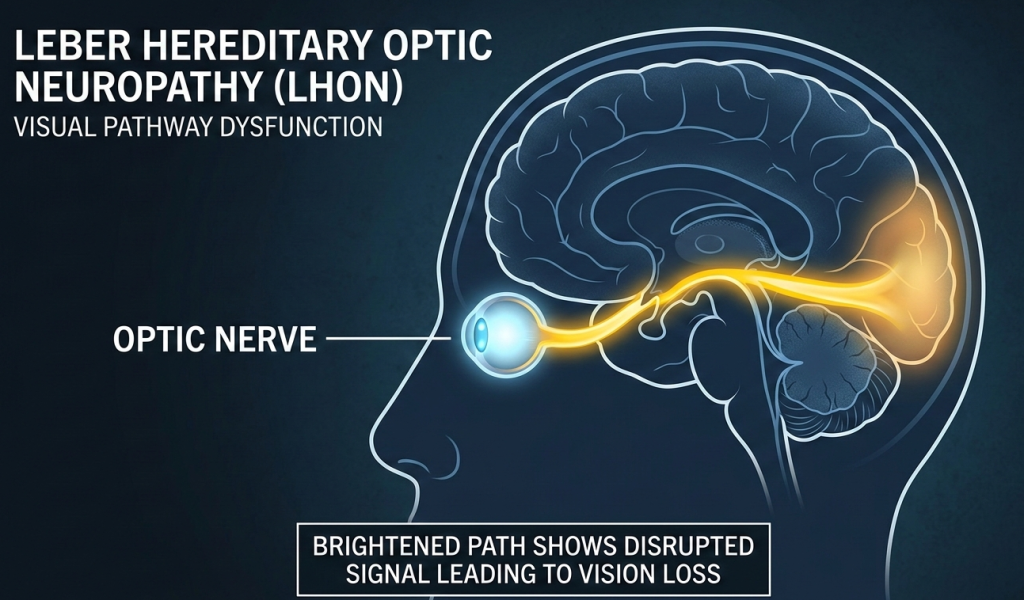
Leber Hereditary Optic Neuropathy is a rare inherited mitochondrial disorder that causes rapid and severe vision loss. The condition occurs when genetic mutations disrupt energy production in retinal ganglion cells, the nerve cells responsible for transmitting visual signals from the eye to the brain.
These cells are particularly vulnerable in the macular region of the retina, which explains the early loss of central vision.
Symptoms typically begin with sudden, painless blurring of vision in one eye, followed by involvement of the second eye within weeks or months. The disease can significantly affect independence, education, and employment due to the loss of central vision required for tasks such as reading and recognizing faces.
LHON is estimated to affect approximately 1 in 15,000 to 50,000 people worldwide and most often occurs in individuals between 15 and 35 years of age. Men are affected three to five times more frequently than women.
Reference
FDA Issues a Complete Response Letter (CRL) Regarding the NDA for Idebenone for Leber Hereditary Optic Neuropathy (LHON) in the United States. https://chiesirarediseases.com/media/20260305-fda-issues-a-complete-response-letter-crl-regarding-the-nda-for-idebenone-for-leber-hereditary-optic-neuropathy-lhon
Study to Assess Efficacy, Safety and Tolerability of Idebenone in the Treatment of Leber’s Hereditary Optic Neuropathy (RHODOS), ClinicalTrials.gov ID NCT00747487, https://clinicaltrials.gov/study/NCT00747487
Study to Assess the Efficacy and Safety of Raxone in LHON Patients (LEROS), ClinicalTrials.gov ID NCT02774005, https://clinicaltrials.gov/study/NCT02774005

About Writer
Sana Jamil Khan, BPharm
She is a pharmacy graduate with a keen interest in clinical research, pharmacovigilance, and medical writing, with a growing focus on publication and scientific content development. In her words, she is passionate about translating complex medical data into clear, evidence-based communication.

